Subproject 7
Dehalogenation of halobenzoates in denitrifying bacteria
Matthias Boll (University of Leipzig)
The anaerobic degradation halogenated aromatics has usually been associated with membrane-bound reductive dehalogenases, corrinoid-containing key enzymes of anaerobic organohalide respiration. In contrast, the complete oxidative degradation of halobenzoates in anaerobes has been reported in denitrifying, sulfate-reducing, Fe(III)-respiring and in anaerobic phototrophic bacteria. The catabolism of halobenzoates is supposed to proceed via CoA esters and includes a so far unknown reductive dehalogenation step. In our project we will study the degradation of various halobenzoates in the denitrifying model organisms Thauera chlorobenzoica and Azoarcus evansii. The major objectives are: (i) determination of the substrate/product of the dehalogenation reactions by CoA-ester metabolome analysis and by the in vitro assays; (ii) characterization of the reductive enzymatic dehalogenation); (iii) characterization of key enzymes involved in halobenzoate catabolism; and (iv) the determination of the minimal enzymatic prerequisites for growth on halobenzoates.
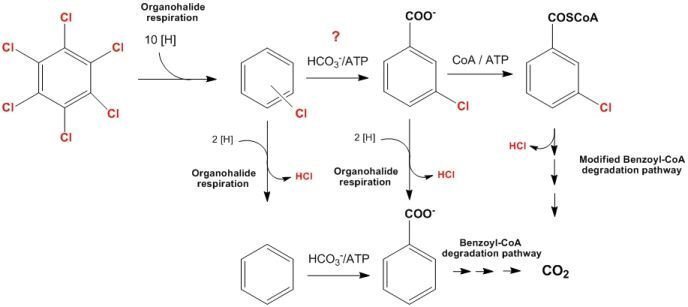
Scheme of haloaromatics dehalogenation and degradation mediated by denitrifying bacteria
Selected References:
Tiedt O, Mergelsberg M, Boll K, Müller M, Adrian L, Jehmlich N, von Bergen M, Boll M.
ATP-Dependent C-F Bond Cleavage Allows the Complete Degradation of 4-Fluoroaromatics without Oxygen
MBio. 2016 Aug 9;7(4). pii: e00990-16. doi: 10.1128/mBio.00990-16.
Kuntze K., Kiefer P., Baumann S., Seifert J., von Bergen M., Vorholt J.A., Boll M.
Enzymes involved in the anaerobic degradation of meta-substituted halobenzoates
Mol. Microbiol. 2011, Nov;82(3):758-69
Kung, J.W., Baumann, S., von Bergen, M., Müller, M., Hagedoorn, P.-L., Hagen, W.R. and Boll, M. (2010) Reversible biological Birch reduction at an extremely low redox potential. J Am Chem Soc 132, 9850-9856.
